AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Henri becquerel atomic theory2/9/2024  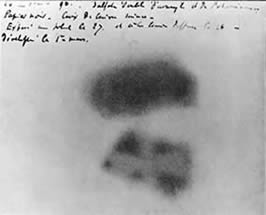
But, he warned, “it is almost a foregone conclusion that similar stores of energy are also possessed by the atoms which … are not radioactive.” That wasn’t scary, Millikan said, because there wasn’t even enough radium in the world to make very much popcorn. Writing in Science News Bulletin (the original Science News precursor) in 1921, physicist Robert Millikan noted that a gram of radium, in the process of disintegrating into lead, emits 300,000 times as much energy as burning a gram of coal. There was no clear clue to how subatomic energy could be tapped for any significant use, military or otherwise. 
In any case, once science began to comprehend the subatomic world, no force could stop the eventual revelation of the atom’s power.īut the path from basic science to the bomb was not straightforward. Radioactivity revealed a new sort of energy, of vast quantity, hidden within the most minuscule components of matter - the parts that made up atoms. Or perhaps it’s better to begin with Henri Becquerel’s discovery of radioactivity in 1896. You could say 1905, when Einstein revealed to the world that E = mc 2. But the science behind nuclear energy originated decades earlier. 
Nuclear fission - the source of the bomb’s energy - was discovered in 1938, less than seven years before Hiroshima. It’s not easy to pinpoint that story’s beginning. So it’s understandable that on the 75th anniversary of the atomic bomb explosion that devastated Hiroshima (August 6, 1945), reflections tend to emphasize the geopolitical dramas during the decades that followed.īut it’s also worth reflecting on the scientific story of how the bombs came to be. But they launched another kind of war, a cold one, that threatened the entire planet with nuclear annihilation. Atomic bombs hastened the end of World War II. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
